Predicting Cancer Deaths With AI! Proposed Predictive Model To Improve End-of-life Prognosis!
3 main points
✔️ As the population ages, end-of-life care for cancer patients - care provided to ensure that they live the remainder of their lives in peace - is attracting increasing attention and survival prediction is becoming more important.
✔️ This study builds a model to predict in-hospital death at the end of life based on the long short-term memory module (LSTM) model, which is based on activity data from wearable devices.
✔️ The assessment utilized the overall prognostic accuracy of the Karnofsky Performance Status-KPS-score, with an outcome score of 0.83
Deep-Learning Approach to Predict Survival Outcomes Using Wearable Actigraphy Device Among End-Stage Cancer Patients
written by
(Submitted on 9 Dec 2021)
Comments: Front Public Health
The images used in this article are from the paper, the introductory slides, or were created based on them.
background
Can wearable devices predict survival?
In this study, we aim to construct a prediction model for in-hospital mortality due to cancer-based on the use of wearable devices and LSTM.
In recent years, survival prediction for an increasing number of cancer patients has gained importance in the clinical practice of end-of-life care - to enable end-of-life preparation, avoid futile medical treatment, and provide optimal palliative care. Various scores have been proposed as prognostic tools for such care: Palliative Prognostic Score-PaP-, Palliative Prognostic Index -PPI-, and others. These scores combine subjective clinical parameters and objective biomarkers, with the patient's performance status (PS) as the main component: the Karnofsky Performance Status (KPS) is used as a tool to assess PS. The Karnofsky Performance Status (KPS) is a tool to assess PS; however, these assessment tools are subjective and require appropriately trained healthcare professionals - thus, challenges such as intra- and inter-observer variability, over- and under-estimation, and so on have been raised. over- and under-assessment, and other issues have been raised.
In this context, wearable devices are attracting attention as an objective evaluation method - these devices constantly record the patient's activity status and can be objectively verified. Using this technology, objective PS assessment of patients with advanced cancer has been reported; however, studies using objective PS data to predict the prognosis of survival have not been reported.
In this study, we objectively measured patients' activity status using a wearable device in patients with terminal cancer - we analyzed the activity data recorded by the wearable device and developed a predictive model suggesting patients' survival outcomes. Furthermore, the prognostic accuracy of the proposed activity monitoring and survival prediction model was compared with the current PS assessment tool, KPS, and also with PPI.
What is a wearable device?
First, a brief description of wearable devices, the subject of this study's analysis, is given.
The wearable device is a generic term for information devices that subjects wear and use. Wearable means wearable or wearable, and there are wristband/wristwatch-type, eyeglass-type, and other types. Since these devices can be worn, they can be used in many aspects of daily life, from exercising to bathing and sleeping - they are thought to be effective in recording exercises such as jogging and swimming, and in acquiring health information by capturing heartbeat, pulse, sleep time, etc. The research team is now working on the development of such a device. In this research, a wristband-type device worn on the wrist was used to acquire data.
technique
In this section, we describe the dataset, the proposed model, and the analysis method used in this study.
data-set
This study was conducted in the Hospice Care Unit of Taipei Medical University Hospital. Patients with terminal illnesses were admitted to the ward for palliative care and management of pain and other symptoms. Clinical data including age, gender, diagnosis, and comorbidities were collected after recruitment, and subjects wore a wristband wearable device on their hands without an intravenous line: the device collected three-dimensional data on hand movement gravity acceleration, angle change, and spin change every second and converted them into three statistical parameters: physical activity, angle and spin The device was worn throughout the hospital stay, except for shower time, as the device was not water-resistant.
Prognostic assessment by KPS and PPI was then performed by two experts: the KPS is a well-established tool designed for PS assessment and assesses normal activity (100) to death (0), taking into account ambulation, activity, evidence of disease, self-care, need for assistance, and disease progression. We also performed the PPI, a composite prognostic tool based on an assessment of PS and other clinical symptoms: the PPI considers PS and clinical symptoms such as oral intake, edema, dyspnea at rest, and delirium to calculate an overall prognosis. Scores range from 0 to 15, with a PPI >6.0 estimated survival of <3 weeks. To ensure interpersonal consistency, all KPS and PPI assessments were performed by the same specialist.
Data preprocessing and LSTM models
The data collected by the wearable device was time series data with three features - physical activity, angle, and spin. The variation in data length was corrected by zero padding up to the maximum length of the time series. To avoid gradient loss, a mean value of 20-time steps was selected and the time series was shortened to < 500-time steps.
This study used LSTM to build a model to predict a patient's clinical condition at discharge - death or discharge in stable condition - see figure below.
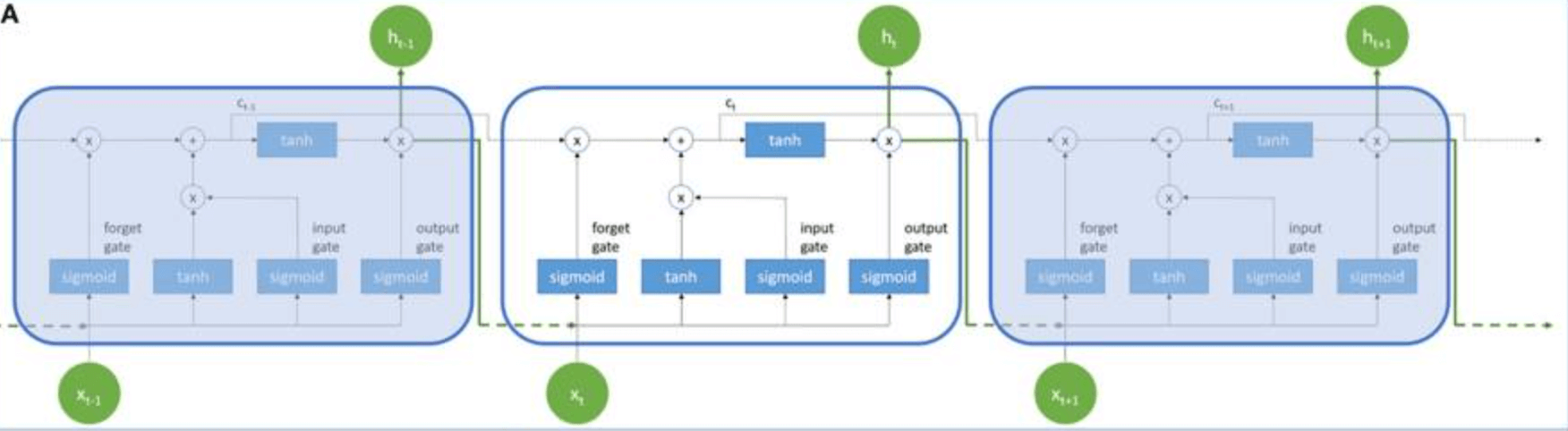
x and h are the input and output values of the LSTM cell, respectively, and the memory cell value of each LSTM cell is c. The subscripts x, h, and c represent different time points. Each LSTM cell has an input gate, an oblivious gate, and an output gate: the input gate determines whether the neuron writes the input value to the memory cell; the output gate determines whether the neuron reads the value in the memory cell. We also utilize hyperbolic tangent function -tanh- and sigmoid function -σ- as activation functions of LSTM. In this study, we built a predictive model using LSTM cells to process 3D time series data - see figure below
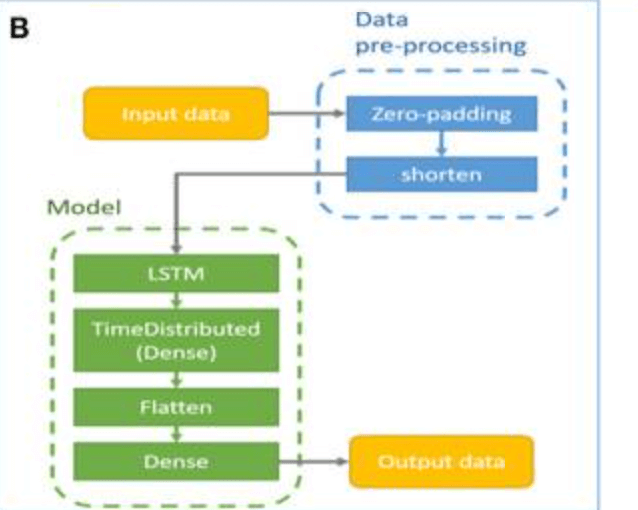
The model consists of an LSTM layer, TimeDistributed layer, etc. to adjust the parameters. The model is designed to generate survival predictions based solely on patient activity data, and patient demographic and clinical data - i.g. comorbidities - are not utilized by the model.
The clinical outcome of participants was determined by death (1) or discharge in stable condition (0) at the end of the hospitalization period. The cut-off values for KPS and PPI were 50% and 6.0, respectively, from previous studies. The predictive accuracy of KPS and PPI was investigated for sensitivity, specificity, overall accuracy, area under the ROC curve -AUC-, etc., were investigated.
result
This section describes the results of the evaluation in this study.
Prognostic accuracy of KPS and PPI
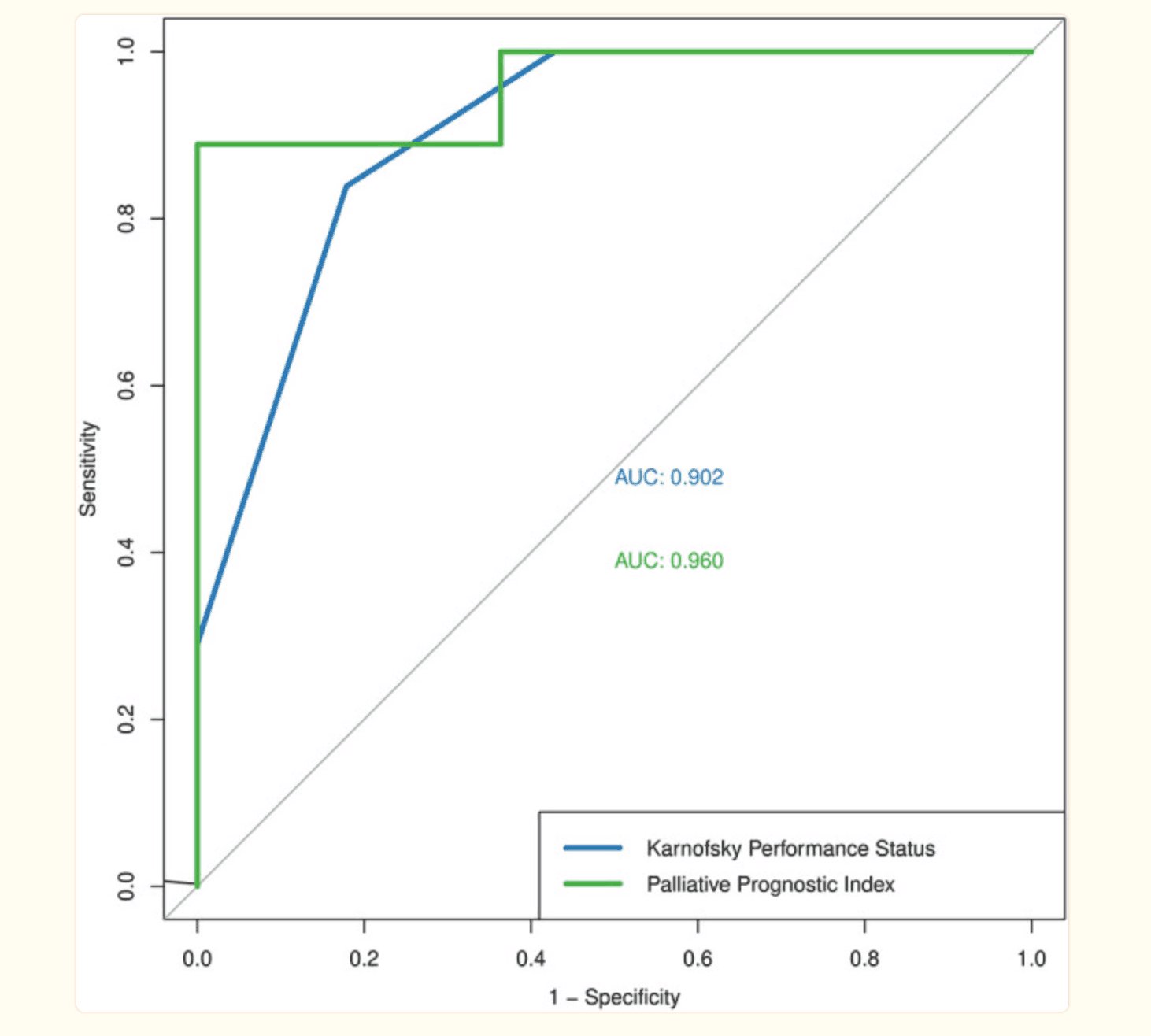
First, the absolute numbers of true positives, false positives, false negatives, and true negatives for the KPS and PPI assessments were defined: true positives were defined as KPS <50% or PPI >6.0 at the baseline visit and death at the end of the hospitalization period. The predictive performance of the KPS score was 83.1% overall predictive accuracy, 82.1% sensitivity, 83.9% specificity, and 0.902 AUC - see figure below. The predictive performance of the PPI score based on binary outcomes was 95.0% overall predictive accuracy, 88.9% sensitivity, and AUC 0.960.
Learning LSTM Survival Prediction Models
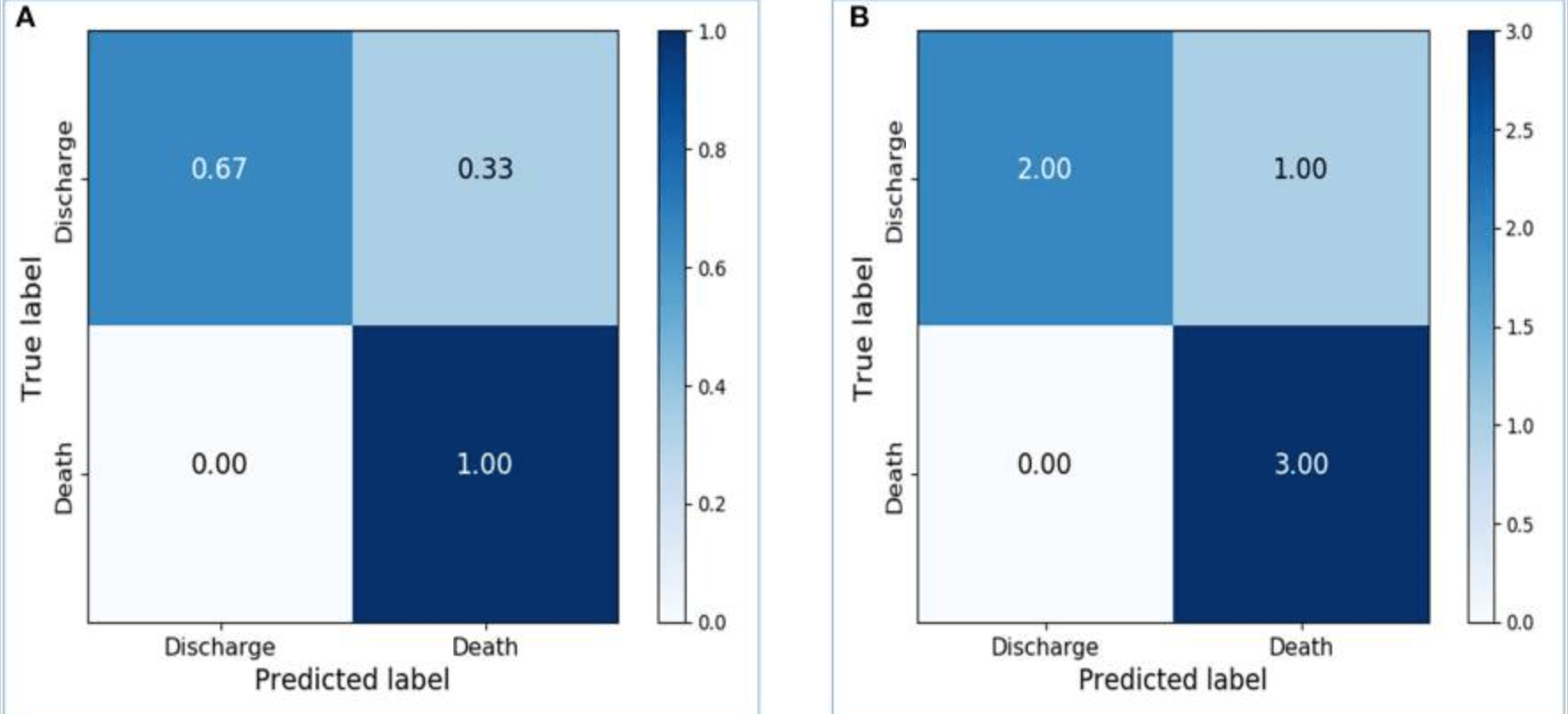
Preliminary models based on activity data recorded within 48 hours of admission resulted in an accuracy of 0.8667 for the training dataset and 0.7143 for the test dataset. The confusion matrix - see figure below - shows the confusion matrix with and without normalization, respectively. The sensitivity, specificity, and AUC of the model in the test data set were 0.8333, 0.625, and 0.7292.
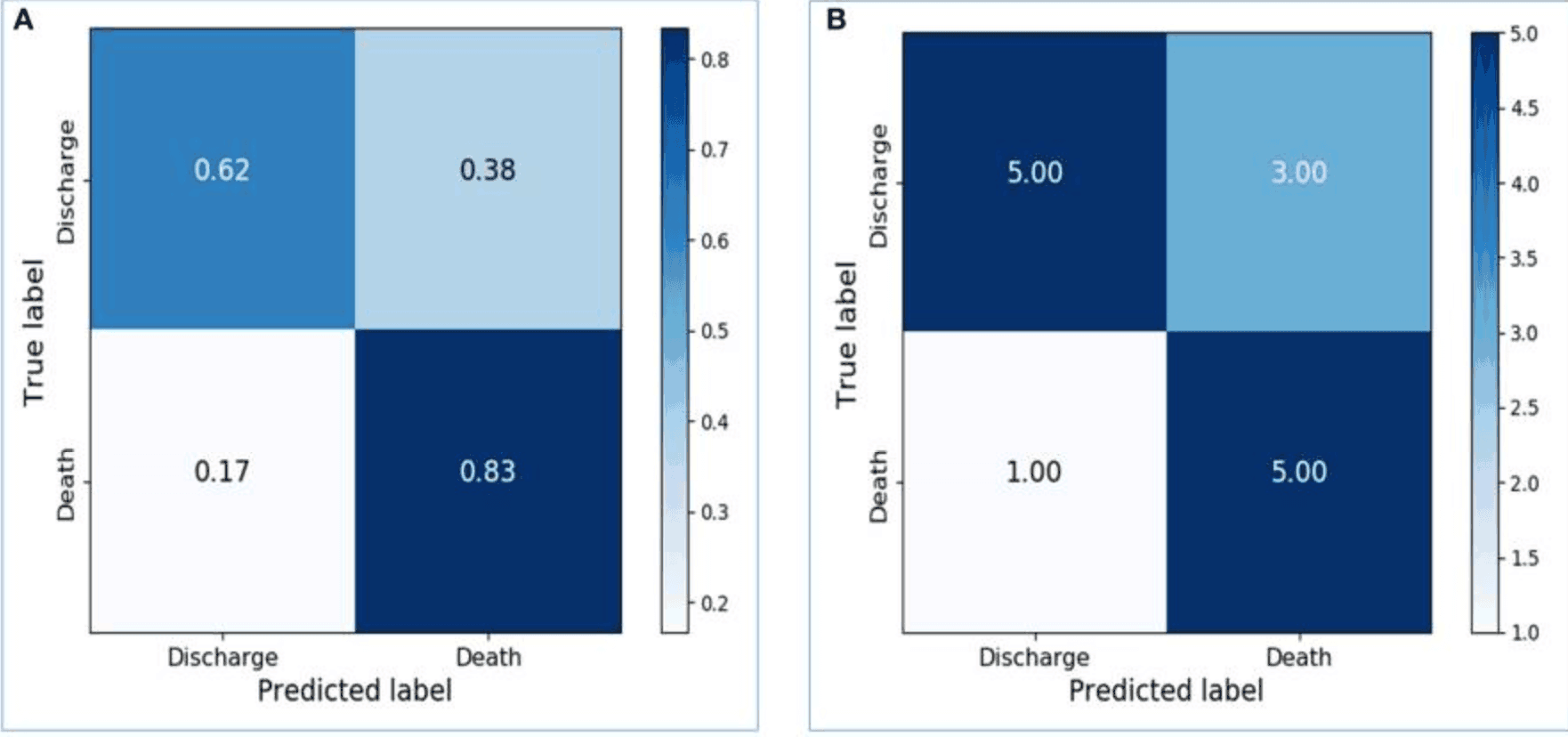
These results demonstrate the feasibility of LSTM in classifying time series data collected by wearable devices without physiological information. Additionally, the dataset was sliced into training, validation, and test data, and the final model was created with appropriate parameters. As a result, the training accuracy increased to 0.9667, and the validation and test accuracies were 0.75 and 0.8333, respectively. Increasing the number of LSTM units from 64 to 256 significantly improved the performance of the model on the test data set: the sensitivity, specificity, and AUC of the model were 1.0, 0.6667, and 0.8333, respectively - see figure below.
consideration
In this study, we proposed a predictive model based on a wearable device to predict survival in patients with terminal cancer - the LSTM was implemented on objective activity data from the wearable device compared to subjective PS assessment, showing high prognostic accuracy. . The wearable device employed in this study was confirmed to be lightweight, inexpensive, and able to provide useful activity data for survival prediction of terminal cancer patients. These results suggest that the introduction of wearable devices and survival prediction models in end-of-life care can facilitate decision-making in clinical practice and better prepare patients for the end of life.
The challenge with traditional prognostic tools - such as PPI - is that they rely on the experience of the clinician; in contrast, the proposed model - activity monitoring and survival prediction model - requires no clinical expertise and uses only a wearable wristband. In contrast, the proposed model - the activity monitoring and survival prediction model - does not require clinical expertise and uses only a wearable wristband; therefore, the proposed model reduces the burden on healthcare professionals in clinical settings and facilitates the possibility of providing end-of-life care in non-hospital settings such as home hospice. It is also inferred that a better prognosis can be achieved by integrating activity assessment and clinical parameters into the survival prediction model.
On the other hand, the challenges in this study may include the following: first, there are data discontinuities - due to the battery charging requirements and the fact that the device is removed during showering - therefore, there is a need to improve the water resistance of the device and introduce better activity tracking devices; and second, it is designed to provide patient outcomes at the end of the hospital stay - i.e., death or discharge in a stable condition. Based on field findings, survival times differ between participants regardless of the final survival outcome -. In contrast, the proposed model only covers binary survival outcomes, not estimated survival times: therefore, additional analyses are needed to include these clinical outcomes.
Categories related to this article



![DrHouse] Diagnostic](https://aisholar.s3.ap-northeast-1.amazonaws.com/media/February2025/drhouse-520x300.png)
![SA-FedLoRA] Communic](https://aisholar.s3.ap-northeast-1.amazonaws.com/media/October2024/sa-fedlora-520x300.png)

![[SpliceBERT] A BERT](https://aisholar.s3.ap-northeast-1.amazonaws.com/media/July2024/splicebert-520x300.png)
![[IGModel] Methodolog](https://aisholar.s3.ap-northeast-1.amazonaws.com/media/July2024/igmodel-520x300.png)